Chemistry: The Mystery and Wonder of Hybrid Orbitals
The Mystery and Wonder of Hybrid Orbitals
In order for the electrons to spread further than 90º from each other, we have to come up with a new model that allows for this.
Up to now, we've learned the shapes and relative energies of s-, p-, d-, and f-orbitals. However, when atoms form covalent compounds, atomic orbitals are insufficient because they force the bonded atoms to be too close to each other. As you might expect, the electrons in covalent bonds, as is the case with electrons everywhere, prefer to be as far apart from each other as possible because they repel each other. Subsequently, all orbitals within an atom that contain valence electrons combine with one another to form "hybrid orbitals."
It's probably easiest to understand how hybrid orbitals work by showing you an example. Let's take a look at the orbital filling diagram for the valence electrons on carbon (see The Modern Atom for more on orbital filling diagrams).
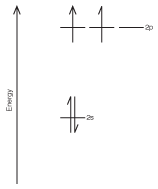
Figure 10.3The orbital filling diagram for the valence electrons on carbon.
As you can see, of the four valence orbitals in carbon, one is filled (2s), two are half-filled (2p), and one is completely empty. This may be the electron configuration of an unbonded carbon atom, but it doesn't explain how carbon can bond four times to form methane. After all, each covalent bond requires the overlap of an orbital containing one electron from each atom. If this model were valid, we would have no bonding with the s-orbital because it's already full, two bonds (one each from two p-orbitals), and one p-orbital that was completely empty. As a result, carbon could only bond twice, a conclusion that doesn't match reality.
Molecular Meanings
Hybrid orbitals are formed by mixing two or more of the outermost orbitals in an atom together. The only element that doesn't form hybrid orbitals is hydrogen, as it has only a single 1s orbital.
What really happens when carbon covalently bonds with other elements is that these four dissimilar s- and p-orbitals mix with one another to form four identical hybridized orbitals. The names of these new hybridized orbitals are a combination of the names of the original four atomic orbitals. In our example, one s-orbital combines with three p-orbitals to form four sp3 orbitals:

Figure 10.4When orbitals combine to form hybrid orbitals, both their shapes and energies are averaged.
As you can see from this diagram, the hybridized orbital configuration of carbon allows room for four covalent bonds, which matches well with the four hydrogen atoms covalently bonded in methane.
As we mentioned in The Modern Atom, s-orbitals are spherical and p-orbitals are at 90-degree angles to one another. As a result, the sp3 orbitals that form when one s- and three p-orbitals combine will be arranged at an angle reflecting a mixture between these two types of orbitals (in this case, 109.5º):
The number of hybrid orbitals that are formed when a covalent molecule bonds depends on the number of single bonds and pairs of unbonded electrons (lone pairs or unshared electron pairs) that are present in the molecule. The electrons in both single bonds and unbonded pairs exist within hybrid orbitals.
The electrons in multiple bonds exist within something called a "π-orbital" that's formed when an unhybridized p-orbital from one atom overlaps with an unhybridized p-orbital from another atom. Let's see how this works in oxygen, O2.
The Mole Says
The electrons on unbonded atoms are located in s-, p-, d-, and f-orbitals. The electrons in atoms that have formed covalent compounds exist within hybrid orbitals.
Each oxygen atom in O2 has two lone pairs of electrons and a double bond with the other oxygen atom. The first of the two bonds between the two oxygen atoms requires hybrid orbitals (because all single bonds require hybrid orbitals), but the second utilizes the spare p-orbital in both atoms. As a result, one s-orbital mixes with two p-orbitals to form three sp2 orbitals.
Let's take a look at what the orbitals on each oxygen atom look like.
Figure 10.7The three sp2 orbitals, which hold the lone pair of electrons and the electrons shared in single bonds, move as far away from one another as possible, at 120º angles. The p-orbitals on each oxygen atom (not shown) overlap to form the double bond.
The three sp2 orbitals spread out as far away from one another as possible due to electron repulsion. The p-orbitals, which are responsible for the double bond, overlap both above and below the bond.
The following table illustrates each type of hybrid orbital that commonly exists in covalent compounds, as well as the name and bond angles of each orbital and the names of each molecular shape.
| Nonhybridized Overlapping Orbitals | Name of Hybrid Orbital | Bond Angle | Molecular Shape |
|---|---|---|---|
| 1s, 1p | sp | 180º | linear |
| 1s, 2p | sp2 | 120º | trigonal planar |
| 1s, 3p | sp3 | 109.5º | tetrahedral |
| 1d, 1s, 3p | dsp3 | 90º, 120º | trigonal bipyramidal |
| 2d, 1s, 3p | d2sp3 | 90º | octahedral |
In common terms, single covalent bonds between two atoms are referred to as "sigma bonds," or ε bonds. These sigma bonds are created by the overlap of two hybrid orbitals. Each multiple bond is referred to as a "pi bond," or πbond. Pi bonds are created by the overlap of non-hybridized p-orbitals. Using the preceding table, each atom in the sp hybridized atom above has two ε bonds and two πbonds.
Excerpted from The Complete Idiot's Guide to Chemistry © 2003 by Ian Guch. All rights reserved including the right of reproduction in whole or in part in any form. Used by arrangement with Alpha Books, a member of Penguin Group (USA) Inc.
To order this book direct from the publisher, visit the Penguin USA website or call 1-800-253-6476. You can also purchase this book at Amazon.com and Barnes & Noble.







