Chemistry: Covalent Compounds Get Mysterious
Covalent Compounds Get Mysterious
Let's recap what we know about how covalent molecules bond:
- When two atoms with similar electronegativities bond with each other, they form covalent compounds.
- Since both atoms want more electrons to be like the nearest noble gas (because of our old friend, the octet rule), neither atom wants to transfer electrons to the other. To form compounds, they need to share electrons instead.
- We also understand a whole bunch of stuff about the properties and naming of covalent compounds.
None of what you've learned so far is wrong. However, your understanding of bonding in covalent compounds isn't yet complete. Specifically, while we know that electrons are shared between two atoms in a covalent molecule, we don't have any ideas about the locations of these electrons. As you might imagine, they're located within orbitals, but what sort of orbitals exist between two atoms?
Why are orbitals important? As it turns out, covalent bonds are formed when two orbitals from different atoms, each of which have one electron in them, overlap so that these two electrons are shared. Because these orbitals need to overlap for a bond to be formed, it's important that we understand the shapes of orbitals that are formed in covalent compounds.
Before we can fully understand the true nature of orbitals in covalent compounds, we must see what's incomplete about our current understanding of orbitals and electrons. Imagine CH4. The type of diagram we saw in Getting to Know Covalent Compounds makes this seem like a simple matter:
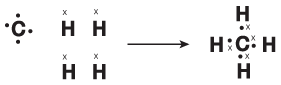
Figure 10.1When one carbon atom bonds with four hydrogen atoms, CH4 is formed.
Though this gives us a nice conceptual view of what's going on, it doesn't do a great job of showing us what happens to the s- and p-orbitals when these compounds bond. Let's examine this in the following figure:
Figure 10.2 almost explains what happens, but it isn't exact because it requires each of the hydrogen atoms on the p-orbitals to be 90º away from each other. Because all electrons have negative charges, they repel each other and prefer to spread out further than 90º. What are we to do?
Excerpted from The Complete Idiot's Guide to Chemistry © 2003 by Ian Guch. All rights reserved including the right of reproduction in whole or in part in any form. Used by arrangement with Alpha Books, a member of Penguin Group (USA) Inc.
To order this book direct from the publisher, visit the Penguin USA website or call 1-800-253-6476. You can also purchase this book at Amazon.com and Barnes & Noble.







