Weather: Feeling the Pressure
Feeling the Pressure
Do you grow tired at the end of the day? I have an excuse for you. Blame it on the atmospheric pressure. If something floats, it's supposed to be lighter than a feather. But the atmosphere is floating and there's nothing feathery about it: Ten to twenty tons of atmosphere rest on the average adult.
Why don't we collapse under such pressure? Fortunately, our bodies exert from within an equal pressure to the atmosphere. So 10 tons of air may be resting on you, but another 10 tons are pushing outward from inside your body. Nobody really collapses and hardly anyone is even aware of the heavy air. But if you had an operation, an injury, or maybe a connective-tissue disease (such as arthritis), your body might not be in exact, natural balance with the surroundings. You really can feel it when the pressure changes within the atmosphere. The following figure shows that balance.
Weather-Wise
The boiling point of water is a function of pressure. At the surface, water boils at 212 degrees Fahrenheit. But at 100,000 feet, water boils at only 40 degrees.
The atmosphere itself is very sensitive to its own variations. A slight change—even 1 percent or less—is enough to get winds blowing and storms coming into circulation.
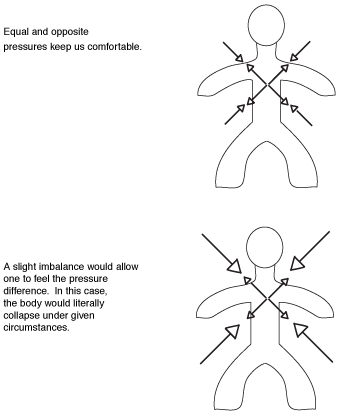
Feeling the pressure.
Origin of Pressure
Because the atmosphere consists of a group of gases, it must have some weight. The following chart shows the composition of the atmosphere—and are we ever lucky! There's just the right mix of oxygen and nitrogen to support life and achieving that mix was no easy matter.
Composition of the atmosphere.
| Permanent Gases | |
|---|---|
| Nitrogen | 78.08% |
| Oxygen | 20.95% |
| Argon | 0.93% |
| Neon | 0.0018% |
| Helium | 0.0005% |
| Methane | 0.0001% |
| Hydrogen | 0.00005% |
| Xenon | 0.000009% |
| Variable Gases | |
| Water vapor | 0-4% |
| Carbon dioxide | 0.034% |
| Ozone | 0.000004% |
| Carbon monoxide | 0.00002% |
| Sulfur dioxide | 0.000001% |
| Nitrogen dioxide | 0.000001% |
| Dust, soot, etc. | 0.00001% |
If the earth were closer to the sun, the intense heat would cause the gases to escape—just like vapor escapes from a boiling pot of water. On Venus, the temperature is as high as 900 degrees. Mercury has an average temperature of 500 degrees. If the earth were smaller, its gravitational pull would be reduced and, like on the Moon, the atmosphere would be lost in space.
Weather-Wise
Temperatures at the equator on Mars are comparable to the temperature found in the polar regions on Earth, but on the Martian poles the temperature plunges to about 180 degrees below 0. That's cold enough to freeze carbon dioxide and form an ice cap of dry ice.
If the earth were larger, lots of nasty gases would stay within the atmosphere because of the stronger gravitational pull. For example, the most common gas in the universe is hydrogen, but our atmosphere doesn't hold much hydrogen. What is present combines with oxygen to simply form low concentrations of water vapor. Hydrogen and many of its compounds are too light to be held down by the earth's gravitational pull. So they escape, which is a good thing, because those hydrogen compounds, such as methane and ammonia, are noxious. (What's more, hydrogen is explosive.) These gases are found around larger planets like Jupiter. That's why life can't be supported there.
So we have the amazing good fortune to have a planet that's just the right size and just the right distance from the sun. This unique arrangement—the combination of distance and size—is also responsible for our weather, including pressure and its variations.
Pressure itself is the weight of a column of air per unit of surface area. It is a force per unit area. That force is a function of the atmosphere's weight relative to the gravitational pull of the planet.
Weather-Speak
A barometer is an instrument used to measure the pressure of the atmosphere. The two kinds of barometers are aneroid and mercurial. The aneroid has a metallic surface and a pointer, with a graduated scale. The mercurial has mercury in a graduated glass tube.
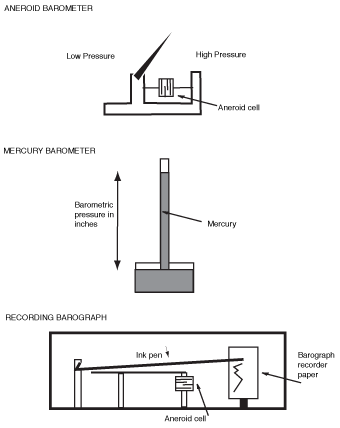
Barometers.
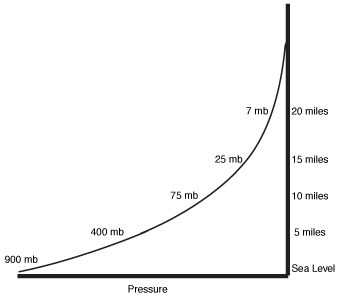
Pressure and elevation.
Normal Variation
Atmospheric pressure is amazingly stable. On the average, the atmosphere weighs about 15 pounds per square inch. Pressure is also expressed in a metric unit, the millibar (mb). The bar is a basic unit of pressure, and a millibar is one one-thousandth of a bar. The average sea-level pressure reading is 1,013.2 millibars. I like millibars, but the most common unit is expressed in inches of mercury, relating to how high a column of mercury will rise within a tube where there's an absence of atmosphere. Normal sea-level pressure comes to 29.92 inches. Those tubes are called barometers.
An aneroid barometer contains no liquid. The term aneroid comes from the Greek meaning "without liquid." A little box within the instrument will expand or contract when the pressure around it decreases or increases. As the volume changes, a dial moves right or left, showing the pressure. This is the type of barometer found in most homes. The pressure reading is also expressed in terms of inches. The figure below illustrate these barometers, which we'll discuss in more detail when we look at the instruments in a weather station.
Generally, at ground level, the atmospheric pressure doesn't fluctuate by more than a few percent—between 30.50 and 29.50 inches. Because the air is rising when it rains, the pressure will be lower. The deepest storm will seldom have a pressure below 29 inches, although some hurricanes can have a pressure below 28 inches. The strongest dry areas of high pressure seldom exceed a pressure of 31 inches. So a relatively small fluctuation of pressure can deliver a hugely different type of weather.
Because pressure results from the earth's gravitational pull, higher elevations will experience reduced pressures—less atmosphere lies above there. The figure at the bottom of the page shows the variation of pressure for different elevations.
By the way, the actual pressure is not as important as the change in pressure. Changes in pressure indicate shifts in vertical motion. Those words around the rim of barometers saying "Clear, Stormy, Rain" don't mean a thing. You don't even have to worry about setting a barometer to a particular standard. Just watch the change, or variation, and the change in pressure will give you solid clues about what is about to occur.
Braving the Elements
The highest pressure ever recorded was 1,085.6 millibars (32.06 inches) at Tosontsengel in NW Mongolia, on December 19, 2001. The temperature was as low as 41 degrees below 0. The extremely cold, dense air contributed to that very high pressure. The lowest sea-level pressure ever recorded was 870 millibars (25.69 inches), on October 12, 1979, about 1,000 miles east of the Philippines within typhoon "Tip." In the United States, the lowest pressure was 26.35 inches in the hurricane of 1935 that crossed the Florida Keys. The absolute lowest pressure has yet to be measured—it would occur within a tornado. The chances of a barometer being in a tornado and withstanding the fury of the storm are slim to none.

Excerpted from The Complete Idiot's Guide to Weather © 2002 by Mel Goldstein, Ph.D.. All rights reserved including the right of reproduction in whole or in part in any form. Used by arrangement with Alpha Books, a member of Penguin Group (USA) Inc.







