Chemistry: The Kinetic Molecular Theory of Gases: Why Gases Do What They Do
The Kinetic Molecular Theory of Gases: Why Gases Do What They Do
As mentioned before, gases are harder to visualize than other phases of matter. This is true not only because it's difficult to see and study them, but because all the molecules in a gas behave independently of one another. As a result, studying the behavior of a gas containing one mole of molecules that are flying all over the place is a lot harder than studying the behavior of a crystal containing a mole of stationary molecules.
Because it's tough to study all of the particles in a gas, scientists have come up with a variety of theories to simplify gases' behavior so they can be more easily understood. Probably the most important of these theories is referred to as the Kinetic Molecular Theory (KMT).
Bad Reactions
All theoretical models (including the KMT) only approximate the behavior of the thing being modeled. The approximations that define each model are designed to make the real phenomenon easier to understand and predict. However, no model is perfect, which explains why weather forecasting models usually get the five-day forecast wrong.
The KMT makes the following assumptions about the behavior of the particles in a gas. Again, these assumptions are not always true, but allow us to understand gases more easily.
The Particles in a Gas Are Infinitely Small
We've mentioned on a number of occasions that atoms and molecules are really, really small (~10-10 m, a unit referred to as an "angstrom"). The Kinetic Molecular Theory not only states that atoms and molecules are small, but says they have no volume at all!
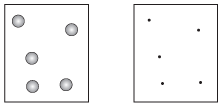
Figure 15.2The molecules in real gases look like the box on the left. The KMT assumes that molecules have no volume at all, like the box on the right. Is this perfectly accurate? No. Is this almost accurate? Yep.
Chemistrivia
Gases are compressible because gas molecules are so small compared to the volume of the gas. If the molecules took up more space, they couldn't squish together as easily.
We make this assumption because it makes the mathematics behind the model easier to follow and because gas molecules are really small. For example, the water molecules in water vapor at 100º C make up approximately 1⁄1700 of the total volume of the gas at standard pressure. Improving the model to account for this very tiny volume wouldn't improve the accuracy of its predictions, but would make the math a lot more difficult. As a result, we just assume that the molecules are infinitely tiny.
The Particles in a Gas Are in Constant Random Motion
The KMT assumes that the particles in a gas, like very small children, constantly move from place to place in an unpredictable fashion. This assumption is correct. Furthermore, the KMT assumes that these particles travel in straight lines until they bash into something, at which point they turn around and go somewhere else. As is also the case with very small children, this assumption is true.
Chemistrivia
The assumption that gas molecules are in constant random motion explains why they have no fixed shape or volume.
Gases Don't Experience Intermolecular Forces
As we mentioned before, gas molecules fly around at very high speeds. Because intermolecular forces are relatively weak, it's rare for molecules traveling past each other at high speeds to interact strongly.
Chemistrivia
Because gases don't experience strong intermolecular forces, all gases are able to mix freely with one another. If intermolecular forces played an important role in gas behavior, polar and nonpolar gases wouldn't be able to mix for the same reason that polar and nonpolar liquids can't mix (see Solutions).
This assumption is similar to my relationship that I had with my downstairs neighbor in college. If I was sitting out on the deck and my neighbor wandered out to his car, his "magnetic personality" (as he referred to it) caused him to stop and speak to me about lobster hunting for hours on end. However, if I was riding my bike in town and I saw him coming the other way, I'd pedal faster and say "Hi-Mikehowsitgoingnicedayseeyouathome-bye!" as I zoomed by. In such instances, my extreme speed kept our interaction to a minimum.
The Kinetic Energies of Gas Molecules Are Directly Proportional to Their Temperatures in Kelvin
The definition of kinetic energy might sound complicated, but what it really means is that when you heat a gas, the molecules move more quickly. Temperature, as it turns out, is a measurement of how much motion the particles in a material have, so it makes sense that increasing the temperature will increase the motion.
Molecular Meanings
Kinetic energy refers to energy caused by the motion of an object. The faster an object moves, the more kinetic energy it has.
The Kelvin scale of temperature is the same as the Celsius scale we've used so far, but it's higher by 273.15º (which we'll abbreviate as 273 for simplicity's sake). We can easily convert degrees Celsius to Kelvin using the following conversion.
- K = ºC + 273
For example, if the temperature outside is 20º C, we can convert this to 293 K. Note that the temperature is properly written simply as "Kelvin" and not "degrees Kelvin."
Chemistrivia
The reason we use Kelvin when working with gases instead of degrees Celsius is that gases often exist at temperatures below 0º C. As a result, if we said that the kinetic energy of a gas was proportional to the temperature in degrees Celsius, the kinetic energy of any gas cooled below the freezing point of water would be negative.
Gas Molecules Undergo Perfectly Elastic Collisions
Elastic collisions are collisions in which kinetic energy is transferred from one thing to another without a loss. If you've ever played pool, you know that the balls slow down just a little bit when they hit the bumpers on the sides of the table. If these collisions were elastic, the balls would bounce off the bumpers moving exactly as quickly as they hit them, never stopping until they either hit a pocket or you got tired of watching them bounce around the table.
Excerpted from The Complete Idiot's Guide to Chemistry © 2003 by Ian Guch. All rights reserved including the right of reproduction in whole or in part in any form. Used by arrangement with Alpha Books, a member of Penguin Group (USA) Inc.
To order this book direct from the publisher, visit the Penguin USA website or call 1-800-253-6476. You can also purchase this book at Amazon.com and Barnes & Noble.







