Chemistry: Separating Mixtures
Separating Mixtures
One of the most important things that chemists do is separate mixtures. For example, when I was working in a chemistry lab and I would go out for lunch, it was of vital interest to me that I effectively separate the pickles from the rest of the hamburger, lest I become sick later in the day. Other chemists I know have also found the ability to separate mixtures to be important when eating trail mix or scraping mud from their shoes. Generally, I've found it's easier to separate the components in a heterogeneous mixture (e.g., the pickles from hamburgers) than the components in a homogeneous mixture (e.g., the rum from a Pia Colada) because it's easier to pick things apart when you can see the different components.
It's good that chemists get so much practice separating mixtures like these in their everyday lives because mixture separation is important for other purposes as well. Let's take a look.
Filtration
One of the simplest methods used to separate mixtures is filtration. If one of the components is a liquid and the other is a solid, filtration is as easy as pouring the whole mixture through filter paper. An everyday example of filtration can be seen in a coffee maker, where the coffee passes through a paper filter but the grounds do not.
Distillation
When one compound is dissolved in another, or when two liquids are mixed together, the most commonly used method to separate them is distillation. In a distillation, the mixture is slowly heated over a Bunsen burner or hotplate. Because the components in a mixture have different boiling points, one of them will boil before the other. The vapor from this compound can be collected from a condenser, enabling it to be isolated in a pure form. A distillation apparatus is shown in the following figure.
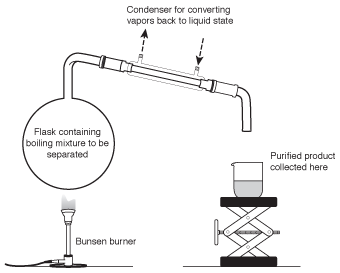
Figure 6.3A distillation apparatus (frequently referred to as a "still") uses the different boiling points of the components in a mixture to separate them.
Chromatography
At some time or another, all of us have marked our shirt with a pen. Sometimes we get lucky and the ink doesn't stick well to the fabric—in these cases, we can clean the shirt by putting it in the wash. Sometimes we get very unlucky and the ink sticks to the fabric so well that it's there for good, no matter how many times it's washed with bleach and detergent.
In the same way, chemical substances can frequently be separated from one another based on how well they stick to a solid. The use of this difference in "stickiness" to separate the components of a mixture is referred to as chromatography.
Typically, chromatography is performed by placing a mixture of two or more chemicals into a glass column filled with silica. When an organic solvent such as ethyl acetate or alcohol is poured through the column, one of the components of the mixture will tend to stick to the silica better than the other. As a result, the less sticky one will pass through the column more quickly, while the stickier one will take a little longer.
Extraction
Let's say that you have a compound dissolved in a liquid that you want to remove. For example, you have a small amount of salt dissolved in oil and want to remove it. How would you do this?
Though distillation could do the job, it takes a long time and considerable effort. An alternate way of making this separation is to find a liquid that isn't soluble with the first liquid and that's better at dissolving the salt than the oil is. When the two liquids are mixed and shaken, the salt will tend to move from the oil (where it's not very soluble) into the water (where it is). When this process is complete, it's a simple matter to pour out the water, leaving behind the pure oil.
Excerpted from The Complete Idiot's Guide to Chemistry © 2003 by Ian Guch. All rights reserved including the right of reproduction in whole or in part in any form. Used by arrangement with Alpha Books, a member of Penguin Group (USA) Inc.
To order this book direct from the publisher, visit the Penguin USA website or call 1-800-253-6476. You can also purchase this book at Amazon.com and Barnes & Noble.







