Anatomy and Physiology: Now I Know My WBCs
Now I Know My WBCs
Although there are many types of white blood cells (see Figure 10.4), they all share a similar function, that of protection from foreign substances, pathogens, and dead cells (they need to be cleared away, after all). Despite their similarities in function, there are differences; let's take a quick look at what they do.

Figure 10.4
Some of the many varieties of white blood cells (WBC). (LifeART©1989-2001, Lippincott Williams & Wilkins)Monocytes do their work outside the blood vessels, although they can move in and out. They survive for a few months, wandering around (fittingly enough) as wandering macrophages, engulfing foreign bodies through phagocytosis. An elevation in the number of these in the blood is a sign of mononucleosis. One cool thing about these guys is what they do when the object they need to engulf is too big for only one to do the job: they call for backup. A bunch of them join up to make a phagocytic giant cell, which then does the job!
Lymphocytes have the longest lifespan of any white cell, living from months to decades! The long lifespan is useful in terms of the B cells, which make antibodies, and can store a memory of the antigen for decades, a fact that makes vaccines possible. I will discuss all of this in more detail when I explain immunity.
Flex Your Muscles
A sign is an objectively measurable indication of swelling, and so on. Don't confuse this with a symptom, which is subjective, such as pain. As of this writing there is no objective measurement for pain.
If you have ever had any swelling, which acts to cushion and protect damaged tissue, basophils were responsible. Basophils respond by leaving the blood vessels nearest the injury and releasing chemicals, including histamines, which induce swelling. Some medicines used to reduce swelling are therefore antihistamines. Neutrophils and eosinophils are similar in that they both act through phagocytosis, and they both release cytotoxic (cell-killing) enzymes, but eosinophils in particular respond by attacking cells and pathogens that have antibodies attached. Although some antibodies can actually destroy a pathogen, most simply label the cells as food for the hungry eosinophils (“Come and get 'em!”). These same eosinophils are also important in getting rid of parasites, and are markers for allergies.
Clots in Store
The last formed elements are the only ones that are not truly cells. Thrombocytes, or platelets, are cell fragments that are essential to the clotting process, as discussed in the next section. The term platelets is more commonly used when referring to human blood, because they are only fragments; vertebrates (craniates?) other then mammals have truly nucleated cells performing this function, and thus making them more deserving of the name thrombocytes.
During blood cell formation, the largest cell is the metamegakaryocyte (sometimes called a megakaryocyte) in the red bone marrow. These cells continuously release cytoplasmic fragments filled with substances essential to blood clotting. The production of so many small platelets in mammals (over the larger thrombocytes of our nonmammalian ancestors) was an increase in clotting efficiency that went hand in hand with the higher metabolism of being mammals. Evolution strikes again!
The contents of platelets are held within a membrane, because if they were released they could trigger part of the clotting process. How the platelets' life-preserving contents—make no mistake here, because without their contents we would quickly lose our life-giving blood with the smallest injury—are actually released, as well as the rest of the clotting process, the sum of which is called hemostasis (prevention of blood loss), is the subject of our next section.
Clots Stem the Tide
Medical Records
Hemophilia is rare genetic disorder in which the cells cannot produce factor VIII. Although only one small part of the clotting process, the absence of that one factor makes the formation of the clot impossible. A chain, after all, is only as strong as its weakest link. Genetically engineered factor VIII has made it possible for hemophiliacs to live fairly normal lives by injecting factor VIII—previously isolated from whole blood—without the risk of blood-borne pathogens.
Exactly how do clots form, and how are they helpful in the healing and repair process? I mentioned the role of the fracture hematoma in the healing of bone in The Bones but clots also form scabs, which keep the injured tissues underneath protected while new cells are made.
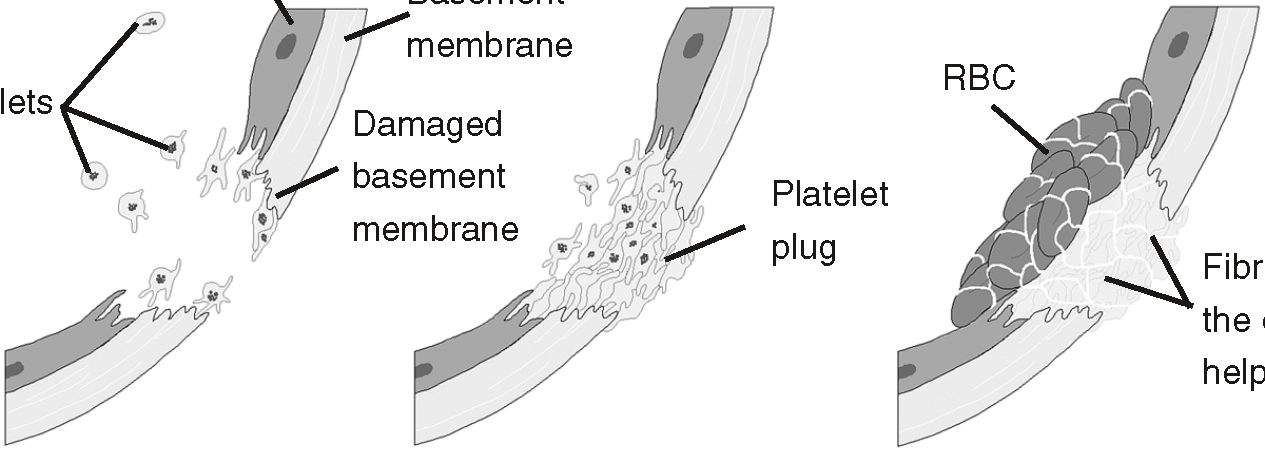
Figure 10.5Without the ability to form clots, our lives are at the mercy of the slightest cut.
First, many chemicals are involved in clotting. Twelve of them are called clotting factors; they are numbered I through XIII because factor VI was found to be the same as factor V. This list of 12 factors is nonetheless incomplete, for other substances, such as ADP and serotonin, are also involved. Rather than tracking down all of these and explaining their function, I will explore the highlights of the three phases of blood clotting: vascular phase, platelet phase, coagulation phase, and clot retraction.
Medical Records
Not all clots function normally. Sometimes a clot continues to grow, extending into the vessel, and ultimately blocking all blood flow through the vessel, instead of just stopping blood flow out. This type of clot is called a thrombus. A clots which breaks loose and gets lodged in a narrower vessel downstream is called an embolus. In both cases, any blood flow beyond either of these abnormal clots is stopped, and the subsequent tissues can die. The severity depends on the area affected.
Hemostasis begins with the vascular phase, a 30-minute period that starts when a blood vessel wall gets damaged. A vascular spasm constricts the vessel walls (vasoconstriction), which will reduce blood loss in larger vessels, but can completely stop capillary blood loss. The initial damage to the walls, coupled with the constriction, causes the exposure of the basement membrane (see The Nervous System), to which platelets attach; the walls also become, for lack of a better word, sticky, which not only helps platelet attachment, but also helps seal off smaller vessels. It is the release of chemicals (including local hormones) by the vessel walls, however, that initiates the second phase, the platelet phase.
The platelet phase starts the attachment of the platelets to the exposed collagen fibers in the damaged vessel walls. Chemicals in the plasma and those released when the platelets themselves break open, stimulate further vasoconstriction, platelet aggregation, as well as the mitosis necessary to, well, repair the walls! Don't forget that all repairs involve mitosis.
The third phase is called the coagulation phase. It is called the third phase because it lasts longer than the platelet phase, but in reality they both start within 30 seconds of the damage to the vessel walls. This phase involves a cascade reaction involving multiple clotting factors, rather like a set of dominoes falling. The most important thing about this step is the chemical conversion (thanks to those clotting factors) of the dissolved fibrinogen in the plasma to a network of fibrin fibers. These fibers trap the RBCs and WBCs and thus stop the blood flow. Refer to Figure 10.5 for an overview of these processes.
The last phase, or clot retraction, pulls the broken edges of the vessel together, which prevents any further bleeding. This retraction makes the damaged area smaller, which makes the mitosis and final repair easier. The only thing left is fibrinolysis, which is the dissolving and breakdown of the clot after the repair is completed.

Excerpted from The Complete Idiot's Guide to Anatomy and Physiology © 2004 by Michael J. Vieira Lazaroff. All rights reserved including the right of reproduction in whole or in part in any form. Used by arrangement with Alpha Books, a member of Penguin Group (USA) Inc.
To order this book direct from the publisher, visit the Penguin USA website or call 1-800-253-6476. You can also purchase this book at Amazon.com and Barnes & Noble.







