Science Projects for Beginners: Chemistry
Chemistry
Salt or Sugar: Which Dissolves Faster in Different Liquids
Solutions are nothing more than mixtures of different compounds or elements. You encounter solutions every day without even realizing it.
Even the air you breathe—which contains water—is a solution of a liquid and a gas. If you drank a soda today, you actually drank of solution of a gas dissolved in flavored water. If you're wearing a bracelet made of sterling silver, you're wearing a solution of two metals.
In this experiment, you'll be working with a liquid solution, which is one of three types of solutions. The other types are gaseous solutions and solid solutions.
So What Seems to Be the Problem?
Basic Elements
A solute is the substance—either a solid, liquid or gas—that gets dissolved. A solvent—which also can be a solid, liquid, or gas—is the substance that does the dissolving. A solution is a uniform mixture of a solute (usually a solid) dissolved in a solvent (usually a liquid).
When you stir a spoonful of sugar into a glass of water, you are forming a solution. This type of liquid solution is composed of a solid solute, which is the sugar, and a liquid solvent, which is the water. As the sugar molecules spread evenly throughout the water, the sugar dissolves.
Mixing a liquid in a gas makes another type of solution, called a gaseous solution. An example of this type of solution is humidity. Humidity is water (a liquid) dissolved in air (a gas).
Standard Procedure
Think about how a sugar cube dissolves in water, compared to a package of loose sugar. The cube dissolves more slowly because fewer sugar molecules are initially in contact with the water.
In a solid solution, such as sterling silver, copper that has been heated at high temperatures is mixed with silver that also has been heated until it melts. The copper is the solute, which is the substance that will dissolve into the solvent. The silver is the solvent.
The type of solution is determined by the state of matter of the solvent. If the substance doing the dissolving is a liquid, the solution is called a liquid solution. If the solvent is a gas, the solution is called a gaseous solution. And you guessed right: A solid solvent will form a solid solution.
Standard Procedure
You can check out the size of salt and sugar crystals under a microscope or magnifying glass, which will allow you to see their shapes, as well. If you draw what you see, using a pencil so that you can illustrate shading, you could include the illustration as part of your final display of your science fair project.
There are a few factors that generally increase the amount of solute that can be dissolved. If you want to dissolve more sugar in the same amount of water, for instance, you could heat the water. You also could grind the sugar into smaller particles to increase its surface area, or you could stir the mixture.
In the years that you've been using salt and sugar on your foods, you've probably noticed that each piece of salt—which actually is a crystal—is a little smaller than each piece of sugar, which also is a crystal.
Basic Elements
A molecule is two or more elements that are chemically combined. A molecule of salt contains sodium and chlorine, which are chemically combined to make sodium chloride. The chemical formula for this salt is NaCl. A molecule of sugar contains carbon, hydrogen, and oxygen. The chemical formula for sugar is C12H22O11.
The problem you'll be attempting to solve in this experiment is whether sugar or salt dissolves faster when mixed into various liquids. Does the size of the pieces affect how quickly they mix with the liquid?
When you dissolve sugar or salt in a liquid—say, water—what happens is that the sugar molecules move to fit themselves between the molecules of water within a glass or beaker. The illustration below shows how the different molecules are arranged in the container.
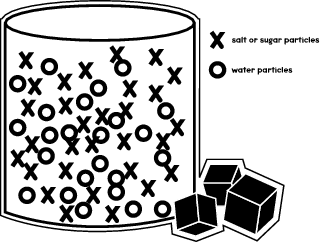
In your experiment, you'll see how salt and sugar molecules move within different liquids and dissolve at different rates.
The title of this section, “Salt or Sugar: Which Dissolves Faster in Different Liquids?” could serve as your project title, if you want. You also could consider one of the following titles for your project:
- The Great Salt vs. Sugar Dissolving Contest
- Using Salt and Sugar to Explore How Substances Dissolve
Whatever name you choose is fine. Let's take a minute now to consider why this project is a valuable use of your time.
What's the Point?
Basic Elements
When there is ample space between the molecules of a solvent, the solvent is said to be unsaturated. When a lot of solute has been dissolved in the solvent, but there is still some space between the molecules, the solution is concentrated. When absolutely no more solute can be dissolved within a solvent, the solution is saturated.
And when the excessive solute has been dissolved by heating the solution, it's said to be supersaturated.
The point of this experiment, in addition to learning whether salt or sugar dissolves faster in various liquids, is to learn how molecules interact in a solution.
As you saw in the preceding illustration, the water molecules take up most of the room in the container. But there is still some available space in which the sugar or salt molecules can fit. Through your experiment, you'll learn how fast the sugar molecules fit into those spaces, as compared to the salt particles.
Knowing this will help you better understand the process that occurs as a substance dissolves.
The control in your experiment will be water. The other liquids in which you dissolve salt and sugar will be the variables.
| Controls: | Solvent—water |
| Solutes—sugar, salt | |
| Variables: | Five different clear liquids (may be colored) |
Remember when you conduct your experiment that it's very important that the liquids you use are all the same temperature. You already learned that sugar dissolves faster in a warm liquid than in a cool one, so you know it wouldn't be an accurate experiment if some of the liquids you use are warm and some are cold. The temperature of the liquid would become a variable.
Therefore, all the liquids you use—including water—should be at room temperature. If you normally keep them in the fridge, be sure to allow them to sit out on the counter overnight until they are all the same temperature.
To give you a little more flexibility when you conduct the experiment, you may choose the liquids in which you'll dissolve sugar and salt. There's no point in having to go out and buy additional liquids if you've already got what you need.
Explosion Ahead
Don't assume that liquids that have been sitting in different areas of your house are the same temperature. A bottle of soda that's been sitting in the garage, for instance, may be several degrees cooler than rubbing alcohol from the bathroom closet, or apple juice from the kitchen pantry. Be sure to have all liquids in the same location in order for them to achieve the same room temperature. If you don't, the results of your experiment won't be valid.
Just make sure you choose liquids that are different from each other in taste, color, odor, and purpose. You'll also need to select those that allow you to observe the salt and sugar as it dissolves. If you use milk or orange juice, for example, you won't be able to watch the salt and sugar dissolve. Some suggestions for liquids to consider are:
- White vinegar
- Club soda
- Ginger ale
- Glass cleaner (such as Windex)
- Lemonade
- Tea or iced tea (each at room temperature)
- Apple juice
- Rubbing alcohol
All of these are commonly found around the house, perhaps saving you a trip to the store.
What Do You Think Will Happen?
Now that you know how solutions are formed and some of the factors that will affect the speed at which the sugar and salt you'll be using will dissolve, you should be able to make a good guess as to which one will dissolve faster.
Standard Procedure
If you haven't done this experiment before, you won't know if the liquids you use will be a factor in dissolving salt and sugar. This makes it more difficult to form a hypothesis, but don't worry. Whether your hypothesis turns out to be correct, or not, does not affect the validity or outcome of your experiment.
While you won't know until after your experiment if properties of the different liquids you choose will affect the rate at which the salt and sugar dissolve, you do know that salt crystals are generally smaller than sugar crystals. And you know that the temperature of the liquids will not be a factor in your experiment.
Just try to use your past experiences, the information you've read earlier in this section, and your common sense to come up with a sound hypothesis.
Remember that your hypothesis must be stated as an objective sentence, not a question. So go ahead and -make your guess as to whether the salt or sugar will dissolve faster, and let's get started with the experiment.
Materials You'll Need for This Project
Some liquids suggested for use in this experiment are white vinegar, club soda, ginger ale, glass cleaner, rubbing alcohol, apple juice, lemonade, and tea. If you want to substitute another liquid for one or more of the ones suggested, that's fine. Just be sure that all liquids are clear and at room temperature.
The amounts of materials listed below are enough for you to conduct the experiment three times with each liquid. You'll need:
- 12 clear, plastic cups (10 ounce [300 ml])
- One permanent marker
- One (1 teaspoon) (5.0 ml) measuring spoon
- One (12 teaspoon) (2.5 ml) measuring spoon
- One (1 cup) (240 ml) measuring cup
- 8 teaspoons (40 ml) salt, divided in 16 (12 teaspoon) portions
- 8 teaspoons (40 ml) sugar, divided in 16 (12 teaspoon) portions
- 48 ounces (1,440 ml) water at room temperature
- 24 ounces (720 ml) each of five different, clear liquids, all at room temperature
- One clock or watch with a second hand
- One clear plastic cup containing eight fluid ounces (240 ml) water at room temperature
Remember to make sure that all liquids are at room temperature.
Conducting Your Experiment
When you've gathered all your materials, you'll be ready to begin your experiment. Just follow these steps:
Standard Procedure
Keep a cup of plain water in sight so you can compare it to the cups containing salt and sugar. It will be interesting to watch how the appearances of the liquids change as the salt and sugar dissolve.
Explosion Ahead
It's going to take a little while for the sugar and salt to dissolve. For best results, do not stir the solutions, as doing so will present an additional variable. If you must stir, then stir each solution three times, and stop. Only stir after you notice there is solute at the bottom of each of the two containers. Stirring the solutions unevenly will cause your experiment to be invalid.
- Using the permanent marker, write “salt” on six of the plastic cups, and “sugar” on the other six.
- Place 1/2 teaspoon (2.5 ml) of salt into each of the six cups labeled “salt.”
- Place 1/2 teaspoon (2.5 ml) of sugar into each of the six cups labeled “sugar.”
- Add 8 ounces (240 ml) water to one cup containing salt, and one cup containing sugar. Immediately record the time at which the water was added on a data chart similar to the one shown in the next section, “Keeping Track of Your Experiment.”
- Observe the solutes (salt and sugar) dissolving in the solvent (water). Record on the data chart the time at which it appears to you that each solute has completely dissolved. These times will probably not be the same.
- Calculate the elapsed time during which the dissolving occurred. Take the time at which the water was added to the cups and the dissolving started, and subtract it from the time the dissolving ended. This gives you the total minutes it took for the salt and sugar to completely dissolve in the liquid.
- Repeat steps 4 through 6, using each different liquid instead of the water.
- Wash, rinse, and thoroughly dry each of the 12 cups.
- Repeat steps 2 through 8 two more times, for a total of three trials for each of the six liquids.
- Calculate an average dissolving time for the salt and the sugar in each of the six liquids.
Remember that to find the average time it took for the salt and sugar to dissolve in each liquid, you add the three times recorded for each one, then divide them by three. The number you get when you divide the times is the average time.
Keeping Track of Your Experiment
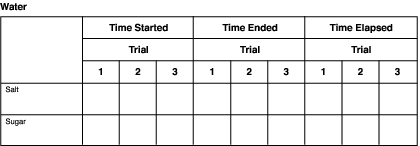
Charts such as the following one can be used to record information for each solvent. Simply change the names of the solvents in the heading.
Be sure to record the times as you go along. Don't depend on your memory to write them down later. You're going to have a lot of numbers by the time you finish your experiment.
Putting It All Together
What did you notice about the rates at which the salt and the sugar dissolved? Did you prove your hypothesis to be correct? Or incorrect? Could you detect any type of pattern as you added the salt and sugar to the various liquids? Was it obvious that the salt dissolved better and faster in some liquids compared to the sugar? Can you think of any reasons for which that might have occurred?
Do you think that the chemical natures of the solute and the solvent affected the dissolving rates? Use the information you gathered when you researched your topic to help you answer these questions.
The more you know about your project, the better able you'll be to analyze your data correctly and come up with a sound conclusion.
Further Investigation
As mentioned earlier, the factors affecting the solubility of solid solutes are:
- Increasing or decreasing the temperature of the solvent
- Increasing the surface area of the solute
- Stirring
If you wanted to take this project a step or two further, you could design an experiment that would test one—or perhaps all—of these variables.
You could easily compare the rate at which sugar cubes dissolve in liquid with the dissolving rate of granulated sugar.
Or you could use the same solute—say, sugar—and test whether stirring the solution caused it to dissolve faster. Heating and cooling the solvent as you add the same solute also would be a possibility for further experimentation.
If you're curious and willing to experiment, you probably can think of many variations for this project. And, because the experiment calls for only common, inexpensive materials, you should be able to experiment to your heart's content.
Excerpted from The Complete Idiot's Guide to Science Fair Projects © 2003 by Nancy K. O'Leary and Susan Shelly. All rights reserved including the right of reproduction in whole or in part in any form. Used by arrangement with Alpha Books, a member of Penguin Group (USA) Inc.
To order this book direct from the publisher, visit the Penguin USA website or call 1-800-253-6476. You can also purchase this book at Amazon.com and Barnes & Noble.







