DK Science: Organic Chemistry
The study of all compounds that contain carbon is called organic chemistry. Carbon atoms are unique. They can combine with each other to make molecules that contain hundreds, even thousands, of carbon atoms. There are more CARBON COMPOUNDS than compounds of all the other elements put together. CARBON TECHNOLOGY uses carbon compounds to make many modern materials, from the interiors of aircraft to medicines.
From butterfly wings to the petals of a flower, all living things are made of carbon compounds. All the processes that happen in living things – such as digestion, movement, and growth – are chemical reactions involving carbon compounds. It is the ability of carbon to make so many different compounds that results in the rich diversity of life on Earth.
Many carbon compounds contain the same few elements, but in different quantities and arranged in different ways. The most important elements to join with carbon are hydrogen, oxygen, and nitrogen. Carbon atoms can form chains of just carbon and hydrogen, which are called hydrocarbons. They can also form rings of carbon, called aromatics.
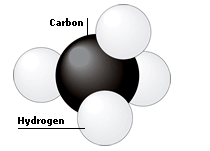
Methane is a hydrocarbon. It contains one carbon atom bonded to four hydrogen atoms. The prefix “meth-” always refers to compounds whose molecules contain only one carbon atom. Methane is a natural gas. It is used in domestic central heating.

Butane gas is a slightly more complex hydrocarbon than methane. Butane contains four carbon atoms and ten hydrogen atoms. The prefix ‘but-’ always refers to compounds whose molecules contain four carbon atoms in a chain.
The carbon industry is one of the largest and most important industries because so many products contain organic (carbon) compounds. Carbon technology is vital to the production of medicines, paints, synthetic fabrics, food flavourings, plastics, cosmetics, and glues. The raw materials that are the basis for these products come from coal, crude oil, and natural gas.

Racing bikes are often made from carbon fibre because it is strong and light, and can be moulded into complex shapes. The carbon fibres are woven into a cloth which is then cut and layered in a mould. The moulded part is filled with a chemical called a resin and then baked in an oven to form the hard, tough carbon fibre material.

New medicines are made to treat specific illnesses by combining organic (carbon) compounds in new ways. Some are similar in structure to compounds found naturally in our bodies or in plants. New medicines undergo a series of tests to ensure they do not have any poisonous effects.

Paint pigments and the dyes that colour our clothes are mostly organic compounds. Pigments coat the surface of a material. Dyes bond with the molecule of the fabric they are colouring. The molecules of pigments and dyes often contain many rings of carbon atoms.

All plastics are organic compounds, made from recycled plastic or from the products of coal, oil, and natural gas. From flexible bags to hard chairs, plastics are light and cheap to make. Their molecules are made of long chains of carbon atoms called polymers.
Polyacrylonitrile (PAN) is heated to 3,000°C (5,432°F) to create thin filaments of carbon fibre. This material is fireproof and five times lighter than steel, yet twice as strong. Carbon fibre has many uses, such as in lightweight sports equipment, car body panels, construction pipes, and on the wings and nose of space shuttles.

